

Variety of bagged media products and ampoules for convenient broth supply and validation processes
Redipor® range of bagged media products
As part of our Redipor® prepared media range we supply bagged media products from 100ml up to 10 litres, with varying fill volumes.
We also supply glass 10ml ampoules.
The use of bagged media products is growing in popularity as they offer a convenient way to supply varying volumes of broth. They are also an important component within operator validation processes.
All batches are supported with QC testing documentation.
Our irradiated Redipor® range of bagged media, offers flexibility and convenience as a solution for various manufacturing applications. These bags have undergone
an irradiation process, which enhances sterility assurance and
reduces the risk of contamination, providing increased confidence in the quality and reliability of results.
Redipor Aseptic Bagged Media Data Sheet
Redipor Irradiated Bagged Media Data Sheet
Redipor Cold Filterable TSB Bag Data Sheet
Safety Data Sheet - Redipor Bottled and Bagged Products

A TITLE THAT WILL DESCRIBE YOUR CONTENT OFFER
WSB - Content Section Ten
The game's not big enough unless it scares you a little. Now we know what they mean by 'advanced' tactical training. A surprise party? Mr. Worf, I hate surprise parties. I would *never* do that to you. Your head is not an artifact!
ANALYTICHEM UK BAGGED MEDIA PRODUCT OPTIONS
Bags and ampoules available: contact us to place your order
Ampoules – 10ml
Our type 1 glass Ampoules are ideal for operator simulations and conveniently supplied in packs of 5.
Each vial has a maximum fill volume of 10ml and a clearly marked break point for snapping open.
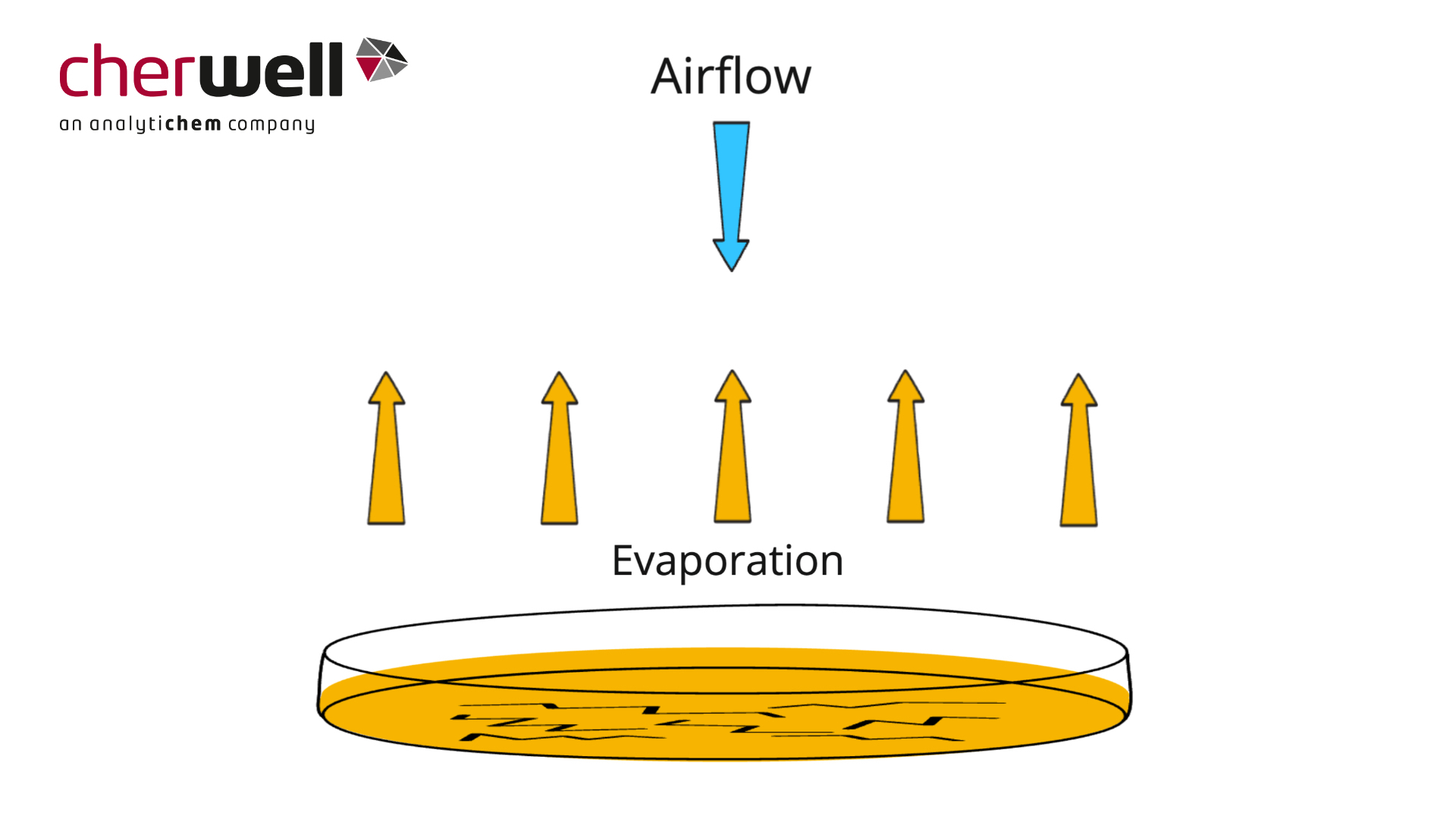
Desiccation of Agar – Choosing Appropriate Media Fill Volume
In this animation Cherwell explains why desiccation and cracking can occur on plates, and why a deeper fill volume may help reduce the impact in your environmental monitoring environment.
What our customers say
Will Brearley, Administrator
Helvic Laboratories
"Alex in sales is fantastic, Hamish has been super helpful too and I've never had a bad experience. Big thanks to Shareen for completing an NC in about 3 hours today too! Saved us a huge headache down the line and we can't thank you all enough. Easily the best supplier we have - there's a reason we rely on you for our most critical media."
Quality Assurance and Cleanroom Specialist
Manufacturer and Supplier of Biocidal and Cleaning Products for Cleanrooms
"Customer service is excellent and I always receive a swift response from one of the team. Should I request a price - I am then provided with information regarding any costs and an estimated delivery date. Once an order is placed, it is always on time. Any queries are quickly resolved. It is a pleasure doing business with Cherwell - it really feels like you care about the customer."
Kieran Broadbridge, Senior Project Leader
Micronclean
"Hassle free. Quality product. Minimal QC issues with good communication when they do occur."
Quality Manager
BioDivide Ltd
"Great customer service and always keeping our company informed of changes particularly concerning regulatory matters that could impact us. Helpful, friendly, efficient."
Technical and Regulatory Affairs Manager
Cleaning Products Supplier
"Thank you! Once again – great service! Goods received within 2 days of ordering and all packaging intact, CoAs received same day."
National Blood Service
Birmingham
"Your service has always been very good, as well as your documentation. Quite a lot of companies you have to chase these days for deliveries and certificates. You always send them promptly."
Julie Bowden, QA Releasing Officer
Pharmacy Manufacturing Unit, Portsmouth
"Friendly, helpful, efficient. Always get a prompt reply to any query. Always feel like a valued customer. Definitely feel like the company cares about it's customers and respects the industry we work in."
Tracey Roberts, Quality Assurance Technician
Wrexham Maelor Hospital
"Cherwell have always provided a great service, with innovative ideas and products. Great customer service too."
Gerald Prout
Kennet Bioservices Limited
"All my contacts with Cherwell have been resolved satisfactorily with 24-48 hours."
Karen Salisbury, Healthcare Scientist
Wrexham Maelor Hospital
"Cherwell provide very efficient service, communication and good product choice."
Microbiology Technician
Industrial Pharmaceutical Company
"The service from Cherwell is excellent, they will always try to provide help where possible."
Jane Maffei, Lead Technician in Aseptic Services
Worcestershire Royal Hospital
"Cherwell are reliable, adaptable, and nothing is to much trouble, excellent customer services. Friendly and helpful."
Head Microbiologist
North Tees and Hartlepool NHS Trust
"Thanks Emma – and all for your help. This does serve to back up the reason we have had such good relations with Cherwell for so many years. Proactive holistic bigger picture thinking."
Louisa Blake, Senior Pharmacy Technician QA/QC
Salisbury District Hospital
"You provide us with an amazing flexible service. Although you are a growing firm you still have the customer service of a dedicated provider! Thanks."
Facilities Coordinator
Roslin Cell Therapies Limited
"Cherwell provide a quick response, very good cooperation and availability, with precise service, high quality reports and overall performance."
need something else?
Browse our products and services catalogue
FREQUENTLY
Asked Questions
Our Redipor Prepared Media range is so extensive that not every product is included in the official price list. In addition to offering such a wide selection of products, we have developed a flexible manufacturing process enabling us to produce bespoke solutions.
Find out more information here.
Contact us to discuss your requirements, whatever they are.
The majority of our prepared media can be stored in ambient conditions, not exceeding 25ºC. There are only a small number of very specialist products that require different storage conditions.
We have never specified refrigeration as a storage condition for our general media as this causes excess condensation and can result in very wet agar, rendering it impossible to use.
View our data sheet on Redipor Storage Conditions.
Settle plates are used to monitor the level of viable particles in the environment through a process of passive air sampling. A viable particle settles on agar plates at a rate dependent on its characteristics and the airflow in the environment.
EU GMP Guide Annex 1 has recommended that 90mm settle plates can be exposed in cleanroom environments for up to 4 hours. However, agar plates may dry out during long exposures where the rate of air exchange is high. So, it might be necessary to use deep filled settle plates, or replace the settle plate after a shorter time to ensure satisfactory growth promotion after exposure.
General purpose media have nutrients that support the growth of most non fastidious culturable microorganisms.
Selective growth media contain components that will inhibit the growth of some types of microorganisms, while supporting the growth of others.
General purpose media, such as Tryptone Soya Agar, are used to produce total counts. While selective media, such as XLD for Salmonella species, are used to test presence/absence of specific types of microorganism.
For Redipor media the Use By date is the last day on which the medium should be inoculated or exposed. Any incubation period that is normal for that product may begin on that day. For example a 14 day sterility test in TSB or a 5 day incubation for a TSA settle plate.
Dip slides can be less sensitive than contact plates or swabs and they often have less stringent QC release criteria. However there are circumstances where they have real advantages.
They are convenient and easy to use by the relatively less skilled.
They are ideal for testing water tanks and hygiene monitoring in food preparation and similarly clean but not aseptic environments. Most importantly they are ideal for field use because they are self contained and the slide can be incubated and assessed without opening the device.
View our data sheet on recycling Redipor products.
ANALYTICHEM UK IS DIFFERENT
Supplying products is only the start
The best products prepared for you
Our Redipor® media products are batch tested, QC certified and delivered when you need them
Bespoke accessories and services
We provide additional equipment or services to make your environmental monitoring more effective
Best customer service
We build long-standing client relationships on understanding, commitment and trust















