
RANGE OF INJECTION VIALS AND DIN BOTTLES FOR STERILITY TEST, OPERATOR AND PROCESS VALIDATIONS
Redipor® range of injection vials and DIN bottles
As part of our Redipor® prepared media range we offer an extensive selection of injection vials and DIN bottles, from 5ml to 500ml. Sterility test broths and rinse solutions available in a choice of fill volumes.
All of these products are fitted with rubber injection stoppers and aluminium tear off seals and are supported with QC testing documentation.
Specialist products are available on request. We will consider any specific requirements and offer solutions wherever possible.
Redipor Bottle Specifications Data Sheet
Safety Data Sheet - Redipor Bottled and Bagged Products

A TITLE THAT WILL DESCRIBE YOUR CONTENT OFFER
WSB - Content Section Ten
The game's not big enough unless it scares you a little. Now we know what they mean by 'advanced' tactical training. A surprise party? Mr. Worf, I hate surprise parties. I would *never* do that to you. Your head is not an artifact!
ANALYTICHEM UK INJECTION VIALS AND DIN BOTTLES
Extensive range of injection vials and DIN bottles: contact us to place your order
Injection vials – 5ml
Injection vials – 10ml
Injection vials – 30ml
Injection vials – 50ml
Injection vials – 100ml
DIN bottles – 100ml
DIN bottles – 250ml
DIN bottles – 500ml
Empty Irradiated Injection Vials
Manufactured in type 1 glass, our empty sterile Injection vials are available in 5, 10, 30, 50 or 100ml capacity and are ideal for process and operator simulations. Supplied with the standard centre tear off seal (CET). An alternative Flip Tear Up seal (FTU) is also available. These vials are supplied in packs of 25.
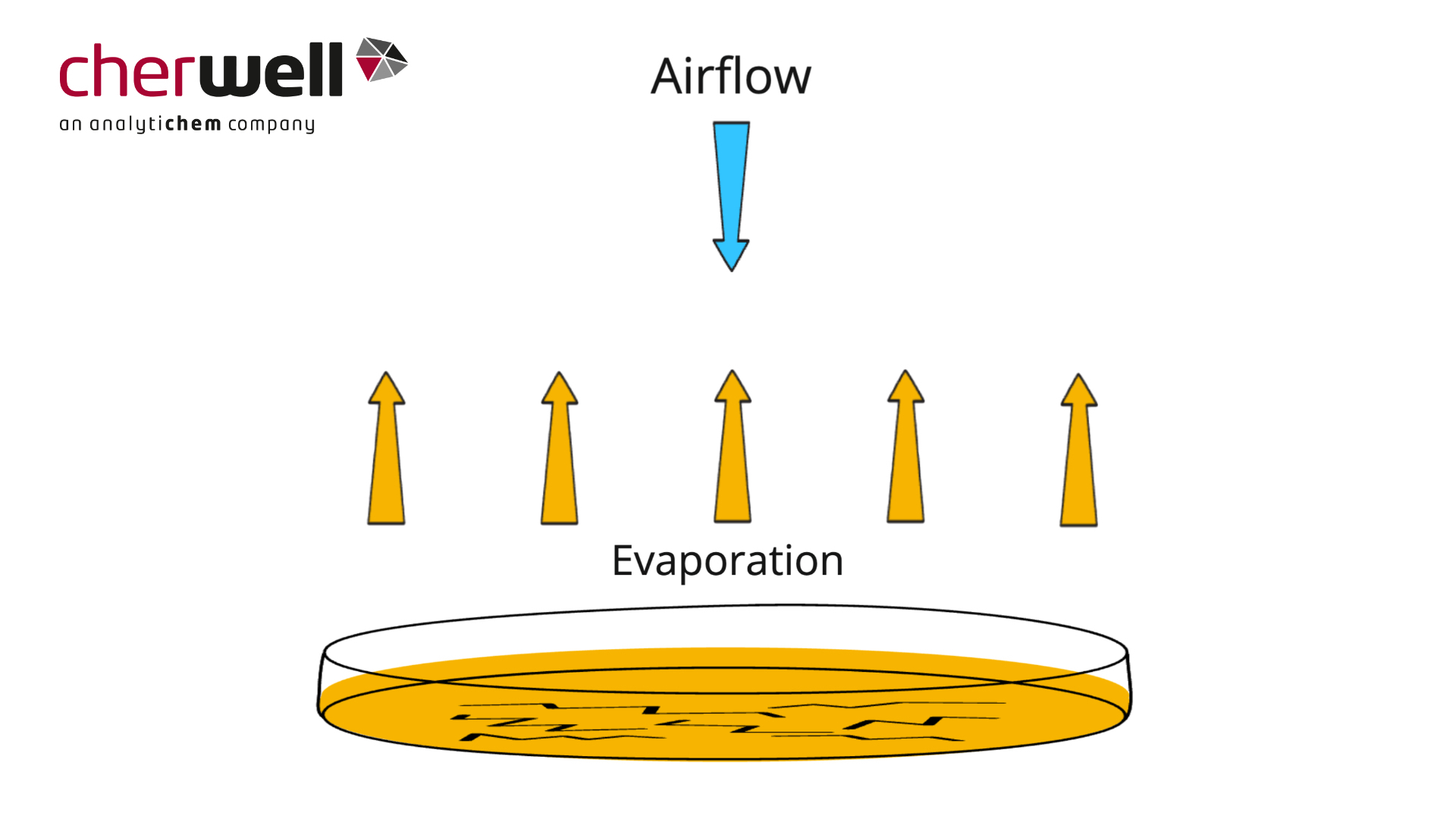
Desiccation of Agar – Choosing Appropriate Media Fill Volume
In this animation Cherwell explains why desiccation and cracking can occur on plates, and why a deeper fill volume may help reduce the impact in your environmental monitoring environment.
What our customers say
Will Brearley, Administrator
Helvic Laboratories
"Alex in sales is fantastic, Hamish has been super helpful too and I've never had a bad experience. Big thanks to Shareen for completing an NC in about 3 hours today too! Saved us a huge headache down the line and we can't thank you all enough. Easily the best supplier we have - there's a reason we rely on you for our most critical media."
Quality Assurance and Cleanroom Specialist
Manufacturer and Supplier of Biocidal and Cleaning Products for Cleanrooms
"Customer service is excellent and I always receive a swift response from one of the team. Should I request a price - I am then provided with information regarding any costs and an estimated delivery date. Once an order is placed, it is always on time. Any queries are quickly resolved. It is a pleasure doing business with Cherwell - it really feels like you care about the customer."
Kieran Broadbridge, Senior Project Leader
Micronclean
"Hassle free. Quality product. Minimal QC issues with good communication when they do occur."
NEED SOMETHING ELSE?
Browse our products and services
FREQUENTLY
Asked Questions
Our Redipor Prepared Media range is so extensive that not every product is included in the official price list. In addition to offering such a wide selection of products, we have developed a flexible manufacturing process enabling us to produce bespoke solutions.
Find out more information here.
Contact us to discuss your requirements, whatever they are.
For Redipor media the Use By date is the last day on which the medium should be inoculated or exposed. Any incubation period that is normal for that product may begin on that day. For example a 14 day sterility test in TSB or a 5 day incubation for a TSA settle plate.
The majority of our prepared media can be stored in ambient conditions, not exceeding 25ºC. There are only a small number of very specialist products that require different storage conditions.
We have never specified refrigeration as a storage condition for our general media as this causes excess condensation and can result in very wet agar, rendering it impossible to use.
Dip slides can be less sensitive than contact plates or swabs and they often have less stringent QC release criteria. However there are circumstances where they have real advantages.
They are convenient and easy to use by the relatively less skilled.
They are ideal for testing water tanks and hygiene monitoring in food preparation and similarly clean but not aseptic environments. Most importantly they are ideal for field use because they are self contained and the slide can be incubated and assessed without opening the device.
View our data sheet on recycling Redipor products.
ANALYTICHEM UK IS DIFFERENT
Supplying products is only the start
The best products prepared for you
Our Redipor® media products are batch tested, QC certified and delivered when you need them
Bespoke accessories and services
We provide additional equipment or services to make your environmental monitoring more effective
Best customer service
We build long-standing client relationships on understanding, commitment and trust















